Liquefied natural gas (LNG) is natural gas (predominantly methane, CH4, with some mixture of ethane, C2H6) that has been cooled down to liquid form for ease and safety of non-pressurized storage or transport. It takes up about 1/600th the volume of natural gas in the gaseous state (at standard conditions for temperature and pressure).
LNG is odourless, colourless, non-toxic and non-corrosive. Hazards include flammability after vaporization into a gaseous state, freezing and asphyxia. The liquefaction process involves the removal of specific components, such as dust, acid gases, helium, water, and heavy hydrocarbons, which could cause difficulty downstream. The natural gas is then condensed into a liquid at close to atmospheric pressure by cooling it to approximately −162 °C (−260 °F); maximum transport pressure is set at around 25 kPa (4 psi) (gauge pressure), which is about one-fourth times the atmospheric pressure at sea level.
The gas extracted from underground hydrocarbon deposits contains a varying mix of hydrocarbon components, which usually includes mainly methane (CH4), along with ethane (C2H6), propane (C3H8) and butane (C4H10). Other gases also occur in natural gas, notably CO2. These gases have wide-ranging boiling points and different heating values, allowing different commercialisation routes and uses. The "acidic" elements such as hydrogen sulphide (H2S) and carbon dioxide (CO2), together with oil, mud, water, and mercury, are removed from the gas to deliver a clean sweetened stream of gas. Failure to remove much or all of such acidic molecules, mercury, and other impurities could result in damage to the equipment. Corrosion of steel pipes and amalgamation of mercury to aluminium within cryogenic heat exchangers could cause expensive damage.
The gas stream is typically separated into the liquefied petroleum fractions (butane and propane), which can be stored in liquid form at relatively low pressure, and the lighter ethane and methane fractions. These lighter fractions of methane and ethane are then liquefied to make up the bulk of LNG that is shipped.
Natural gas was considered during the 20th century to be economically unimportant wherever gas-producing oil or gas fields were distant from gas pipelines or located in offshore locations where pipelines were not viable. In the past, this usually meant that natural gas produced was typically flared, especially since, unlike oil, no viable method for natural gas storage or transport existed other than compressed gas pipelines to end users of the same gas. Natural gas markets were historically entirely local, and any production had to be consumed within the local or regional network.
Developments in production processes, cryogenic storage, and transportation effectively created the tools required to commercialize natural gas into a global market which now competes with other fuels. Furthermore, the development of LNG storage also introduced reliability in previously thought impossible networks. Given that storage of other fuels is relatively easily secured using simple tanks, a supply for several months could be kept in storage. With the advent of large-scale cryogenic storage, it became possible to create long-term gas storage reserves. These reserves of liquefied gas could be deployed at a moment's notice through regasification processes, and today are the primary means for networks to handle local peak shaving requirements.

Specific energy content and energy density
The heating value depends on the source of gas used and the process used to liquefy the gas. The range of heating value can span ±10 to 15 per cent. A typical value of the higher heating value of LNG is approximately 50 MJ/kg or 21,500 BTU/lb.[2] A typical value of the lower heating value of LNG is 45 MJ/kg or 19,350 BTU/lb.
For the purpose of comparison of different fuels, the heating value may be expressed in terms of energy per volume, known as the energy density expressed in MJ/litre. The density of LNG is roughly 0.41 kg/litre to 0.5 kg/litre, depending on temperature, pressure, and composition compared to water at 1.0 kg/litre. Using the median value of 0.45 kg/litre, the typical energy density values are 22.5 MJ/litre (based on higher heating value) or 20.3 MJ/litre (based on lower heating value).
The volumetric energy density of LNG is approximately 2.4 times that of compressed natural gas (CNG), which makes it economical to transport natural gas by ship in the form of LNG. The energy density of LNG is comparable to propane and ethanol but is only 60 per cent that of diesel and 70 per cent that of gasoline.
History
Experiments on the properties of gases started early in the seventeenth century. By the middle of the seventeenth century Robert Boyle had derived the inverse relationship between the pressure and the volume of gases. At about the same time, Guillaume Amontons started looking into temperature effects on gas. Various gas experiments continued for the next 200 years. During that time there were efforts to liquefy gases. Many new facts about the nature of gases were discovered. For example, early in the nineteenth century, Cagniard de la Tour showed there was a temperature above which gas could not be liquefied. In the mid to late nineteenth century, there was a significant push to liquefy all gases. A number of scientists including Michael Faraday, James Joule, and William Thomson (Lord Kelvin) did experiments in this area. In 1886 Karol Olszewski liquefied methane, the primary constituent of natural gas. By 1900 all gases had been liquefied except helium, which was liquefied in 1908.
The first large-scale liquefaction of natural gas in the U.S. was in 1918 when the U.S. government liquefied natural gas to extract helium, a small component of some natural gas. This helium was intended for use in British dirigibles for World War I. The liquid natural gas (LNG) was not stored but regasified and immediately put into the gas mains.
The fundamental patents concerning natural gas liquefaction date from 1915 to the mid-1930s. In 1915 Godfrey Cabot patented a method for storing liquid gases at very low temperatures. It consisted of a Thermos bottle-type design which included a cold inner tank within an outer tank; the tanks being separated by insulation. In 1937 Lee Twomey received patents for a process for large-scale liquefaction of natural gas. The intention was to store natural gas as a liquid so it could be used for shaving peak energy loads during cold snaps. Because of large volumes, it is not practical to store natural gas, as a gas, near atmospheric pressure. However, when liquefied, it can be stored in a volume 1/600th as significant. This is a practical way to store it, but the gas must be kept at −260 °F (−162 °C).
There are two processes for liquefying natural gas in large quantities. The first is the cascade process, in which the natural gas is cooled by another gas which in turn has been cooled by another gas, hence named the "cascade" process. There are usually two cascade cycles before the liquid natural gas cycle. The other method is the Linde process, with a variation of the Linde process called the Claude process, which is sometimes used. In this process, the gas is cooled regeneratively by continually passing and expanding it through an orifice until it is cooled to temperatures at which it liquefies. This process was developed by James Joule and William Thomson and is known as the Joule–Thomson effect. Lee Twomey used the cascade process for his patents.
Commercial operations in the United States


The East Ohio Gas Company built a full-scale commercial LNG plant in Cleveland, Ohio, in 1940 just after a successful pilot plant built by its sister company, Hope Natural Gas Company of West Virginia. This was the first such plant in the world. Originally it had three spheres, approximately 63 feet in diameter containing LNG at −260 °F. Each sphere held about 50 million cubic feet of natural gas. A fourth tank, a cylinder, was added in 1942. It had an equivalent capacity of 100 million cubic feet of gas. The plant operated successfully for three years. The stored gas was regasified and put into the mains when cold snaps hit and extra capacity was needed. This precluded the denial of gas to some customers during a cold snap.
The Cleveland plant failed on October 20, 1944, when the cylindrical tank ruptured, spilling thousands of LNG over the plant and nearby neighbourhood. The gas evaporated and caught fire, which caused 130 fatalities.[6] The fire delayed further implementation of LNG facilities for several years. However, over the next 15 years new research on low-temperature alloys, and better insulation materials, set the stage for a revival of the industry. It restarted in 1959 when a U.S. World War II Liberty ship, the Methane Pioneer, converted to carry LNG, made a delivery of LNG from the U.S. Gulf Coast to energy-starved Great Britain. In June 1964, the world's first purpose-built LNG carrier, the Methane Princess, entered service.[7] Soon after that, a large natural gas field was discovered in Algeria. International trade in LNG quickly followed as LNG was shipped to France and Great Britain from the Algerian fields. One more essential attribute of LNG had now been exploited. Once natural gas was liquefied it could not only be stored more quickly but it could be transported. Thus energy could now be shipped over the oceans via LNG the same way it was shipped in the form of oil.
The US LNG industry restarted in 1965 when a series of new plants were built in the U.S. The building continued through the 1970s. These plants were not only used for peak-shaving, as in Cleveland but also for base-load supplies for places that never had natural gas before this. A number of import facilities were built on the East Coast in anticipation of the need to import energy via LNG. However, a recent boom in U.S. natural gas production (2010–2014), enabled by hydraulic fracturing ("fracking"), has many of these import facilities being considered export facilities. The first U.S. LNG export was completed in early 2016.[8]
LNG life-cycle
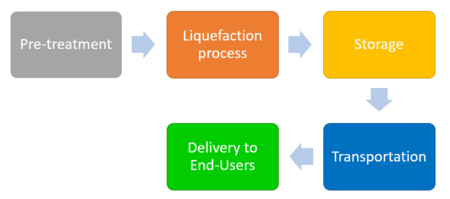
The process begins with the pre-treatment of a feedstock of natural gas entering the system to remove impurities such as H2S, CO2, H2O, mercury and higher-chained hydrocarbons. Feedstock gas then enters the liquefaction unit where it is cooled to between -145 °C and -163 °C Although the type or number of heating cycles and/or refrigerants used may vary based on the technology, the basic process involves circulating the gas through an aluminium tube coils and exposure to a compressed refrigerant. As the refrigerant is vaporized, the heat transfer causes the gas in the coils to cool. The LNG is then stored in a specialized double-walled insulated tank at atmospheric pressure ready to be transported to its final destination.
Most domestic LNG is transported by land via truck/trailer designed for cryogenic temperatures. Intercontinental LNG transport travels by special tanker ships. LNG transport tanks comprise internal steel or aluminium compartment and an external carbon or steel compartment with a vacuum system in between to reduce the amount of heat transfer. Once on site, the LNG must be stored in vacuum-insulated or flat-bottom storage tanks. When ready for distribution, the LNG enters a regasification facility where it is pumped into a vaporizer and heated back into gaseous form. The gas then enters the gas pipeline distribution system and is delivered to the end user.
Production
The natural gas fed into the LNG plant will be treated to remove water, hydrogen sulfide, carbon dioxide, benzene and other components that will freeze under the low temperatures needed for storage or be destructive to the liquefaction facility. LNG typically contains more than 90% methane. It also contains small amounts of ethane, propane, butane, some heavier alkanes, and nitrogen. The purification process can be designed to give almost 100% methane. One of the risks of LNG is a rapid phase transition explosion (RPT), which occurs when cold LNG comes into contact with water.
The most critical infrastructure needed for LNG production and transportation is an LNG plant consisting of one or more LNG trains, each of which is an independent unit for gas liquefaction and purification. A typical train consists of a compression area, a propane condenser area, and methane and ethane areas.
Qatar's giant LNG train is in operation, with a total production capacity of 7.8 million tonnes per annum (MTPA). LNG is loaded onto ships and delivered to a regasification terminal, where the LNG is allowed to expand and reconvert into gas. Regasification terminals are usually connected to a storage and pipeline distribution network to distribute natural gas to local distribution companies (LDCs) or independent power plants (IPPs).
